
Cold Chain Monitoring & Assurance
From production to delivery — prove your cold chain is controlled
A single temperature excursion can destroy a batch of pharmaceuticals, trigger a food safety recall, or invalidate an export shipment. Most cold chain failures aren't caused by equipment breakdown — they're caused by gaps in process, monitoring, and response. Synergic Technologies combines supply chain consulting expertise with Dickson environmental monitoring technology to deliver cold chain assurance that is continuous, compliant, and auditable.
We don't just install sensors. We design the cold chain process, deploy the right monitoring technology, and help you prove compliance to regulators, customers, and export markets.
Why Cold Chain Failures Happen
Cold chain failures are rarely a single catastrophic event. They're the accumulation of gaps — in process, monitoring, and response — that go undetected until product is lost or compliance is questioned.
Excursions Discovered Too Late
A temperature breach during overnight transport or weekend storage isn’t detected until product is already compromised. Manual checks and delayed BMS alerts mean the window for corrective action has closed before anyone knows there’s a problem.
Compliance Gaps You Can’t See
Your cold chain looks compliant on paper — but auditors look at the gaps: missing records between checks, no proof of corrective action, no chain of custody from production to delivery. The absence of evidence is evidence of absence.
Manual Monitoring Can’t Scale
Clipboard checks, standalone data loggers, and disconnected spreadsheets break down as your cold chain grows. More sites, more SKUs, more export markets — the manual approach that worked for one facility doesn’t work for five.
What We Deliver
Cold chain assurance requires both the right process and the right technology. Synergic bridges supply chain consulting and environmental monitoring technology — so you get a solution that is designed for your operations, not a generic sensor deployment.
Consulting — Process & Compliance
Cold Chain Process Design
Map your end-to-end cold chain — production, storage, transport, last mile — and design temperature management processes that are compliant, efficient, and auditable.
Risk Assessment & Gap Analysis
Identify where your cold chain is most vulnerable: handoff points, seasonal variation, equipment failure, human error. Prioritise by risk and business impact.
Compliance Framework Alignment
Align your cold chain processes and documentation with the specific regulatory frameworks that apply to your industry — Medsafe, MPI, NZ Food Act, FSANZ, 21 CFR Part 11, PIC/S, WHO GDP.
Monitoring Strategy & Specification
Define what to monitor, where, how often, and what alarm thresholds and escalation paths to set. Technology-informed but vendor-neutral at the consulting stage.
Technology — Monitoring & Tracking
Dickson Environmental Monitoring
Wireless data loggers, LoRaWAN gateways, and the OCEAView platform for continuous, compliance-grade temperature and humidity monitoring across your cold chain. 24/7 alerting with escalation.
RFID Cold Chain Tracking
Combine environmental monitoring with RFID for product-level traceability through the cold chain — linking temperature records to specific batches, pallets, or shipments for full chain of custody.
3D Temperature Mapping
Qualify storage areas with compliant 3D temperature mapping studies. Dickson’s Mapping Suite enables unlimited in-house studies — meeting WHO, PIC/S, and EU GMP Annex 15 requirements.
Dickson cold chain technology

Cold chain monitoring
OCEAView on tablet — real-time visibility across the cold chain

3D temperature mapping
Mapping Suite — qualify cold rooms to WHO, PIC/S, and EU GMP
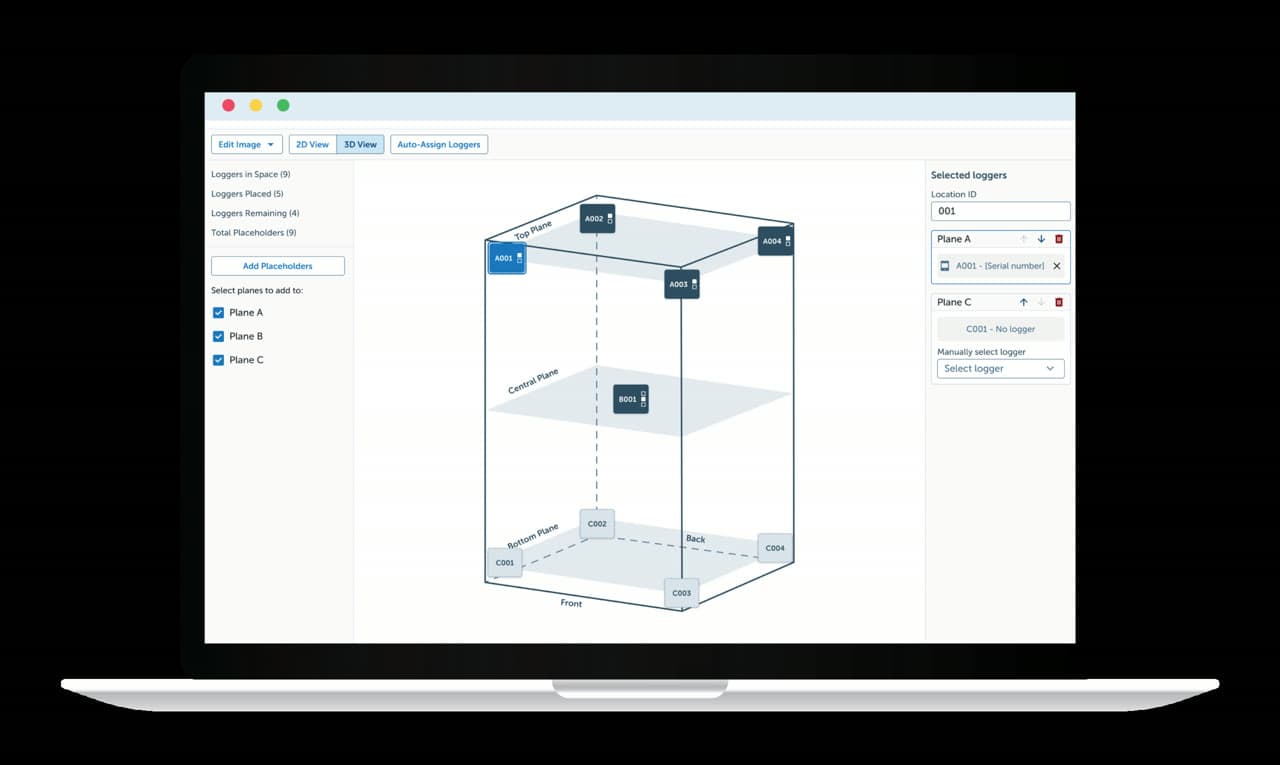
Mapping Suite software
3D logger placement, automated reports, unlimited in-house studies
Compliance Frameworks
Cold chain compliance requirements vary by industry and export market. We align your processes and monitoring to the specific standards that apply to your business.
| Standard | Jurisdiction | Cold Chain Relevance |
|---|---|---|
| 21 CFR Part 11 | USA (FDA) | Electronic records and signatures for pharmaceutical cold chain documentation. Required for NZ pharma exporters to the US market. |
| Medsafe / PIC/S | New Zealand | NZ pharmaceutical regulatory framework aligned with PIC/S PE 009 and EU GMP. Requires validated storage conditions and continuous monitoring. |
| NZ Food Act 2014 / MPI | New Zealand | Requires demonstrable temperature control under Food Control Plans. Applies to all NZ food businesses in the cold chain. |
| FSANZ Standard 3.2.2A | NZ / Australia | Food safety management plans including temperature monitoring. In effect from December 2023 for NZ and Australian food businesses. |
| WHO GDP / TRS 961 Annex 9 | Global | Good Distribution Practice for pharmaceuticals. Explicitly requires temperature mapping and continuous monitoring during storage and transport. |
Industries
Pharmaceutical
Manufacturing, warehousing, distribution, and export cold chain. Stability chambers, cold rooms, transport validation. Medsafe, PIC/S, and FDA compliance for local and export markets.
Food & Beverage
Cold storage, processing facilities, chilled and frozen distribution. NZ Food Act compliance, MPI export requirements, and FSANZ food safety management plans.
Healthcare
Hospital pharmacy medication storage, vaccine cold chain, blood bank monitoring, laboratory freezers. Automated alerting to clinical staff — not facilities teams.
Primary Sector & Export
Dairy, meat, seafood, and horticulture export cold chain. Continuous temperature records from packhouse through port to destination — supporting market access and customer confidence.

Primary sector export — maintaining the cold chain from catch to market
How We Work
Every cold chain engagement follows Synergic's ALIGN → PILOT → SCALE-UP methodology — starting with understanding, proving value on a contained scope, then extending across the operation.
Assess & Design
Map your current cold chain, assess risks, identify compliance gaps, and design the target-state process. Define monitoring requirements and technology specifications.
Prove It Works
Deploy monitoring on a priority site or cold chain segment. Validate processes, alarm thresholds, and escalation paths against real-world conditions before full rollout.
Roll Out & Optimise
Extend across all sites, routes, and products. Embed cold chain monitoring into standard operating procedures. Continuous improvement based on data trends and exception analysis.
Related Capabilities
Cold chain assurance connects to several other areas of our practice. Explore the technology and consulting capabilities that support a complete cold chain solution.
Environmental Monitoring
Dickson monitoring technology — data loggers, sensors, OCEAView platform, and 3D temperature mapping.
RFID Solutions
Product-level tracking and traceability through the cold chain — linking temperature records to batches and shipments.
Supply Chain Consulting
Our full consulting practice — maturity assessment, S&OP, inventory optimisation, visibility, and more.
Environmental Monitoring in the NZ Cold Chain
Why NZ's FMCG and pharmaceutical cold chains need real-time monitoring — and how to select the right solution.
Read article →Frequently Asked Questions
What is cold chain assurance?+
Cold chain assurance is the combination of processes, technology, and compliance frameworks that guarantee temperature-sensitive products remain within specification from production through to delivery. It goes beyond monitoring — it includes process design, risk assessment, corrective action procedures, documentation, and regulatory compliance. Synergic Technologies delivers cold chain assurance by combining supply chain consulting expertise with Dickson’s environmental monitoring technology.
What is the difference between cold chain monitoring and cold chain assurance?+
Cold chain monitoring is the technology layer — sensors, data loggers, dashboards, and alerts that track temperature in real time. Cold chain assurance is the complete system: the monitoring technology plus the processes, compliance documentation, risk management, and corrective action procedures that prove your cold chain is controlled end-to-end. Monitoring tells you the temperature. Assurance proves your products are safe.
Which compliance standards apply to cold chain in New Zealand?+
It depends on your industry. Pharmaceutical cold chains must comply with Medsafe requirements (aligned with PIC/S and EU GMP), plus FDA 21 CFR Part 11 for US exports. Food businesses must comply with the NZ Food Act 2014, MPI requirements, and FSANZ Standard 3.2.2A. Healthcare facilities follow Medsafe guidelines for medication storage. Export cold chains must also meet destination-market requirements — EU GDP, WHO TRS 961, or FDA FSMA depending on the market.
How does cold chain consulting work with environmental monitoring technology?+
The consulting engagement designs the cold chain process — what needs to be monitored, where, what the alarm thresholds should be, and how excursions are managed. The technology (Dickson environmental monitoring, RFID tracking) implements that design. Synergic delivers both: the supply chain consulting expertise to design the right process, and the technology capability to deploy and support the monitoring solution.
Can you monitor temperature during transport, not just in storage?+
Yes. Dickson’s Emerald mobile data loggers and Atlas transport sensors provide continuous temperature recording during transit. For product-level traceability through the cold chain — linking temperature records to specific batches or shipments — we combine Dickson monitoring with RFID tracking. This gives you full chain of custody from production through storage, transport, and delivery.
Ready to Secure Your Cold Chain?
Whether you need to close compliance gaps, replace manual monitoring, or design a cold chain process for a new facility — talk to our team about what you're trying to achieve.